Assay Principle:
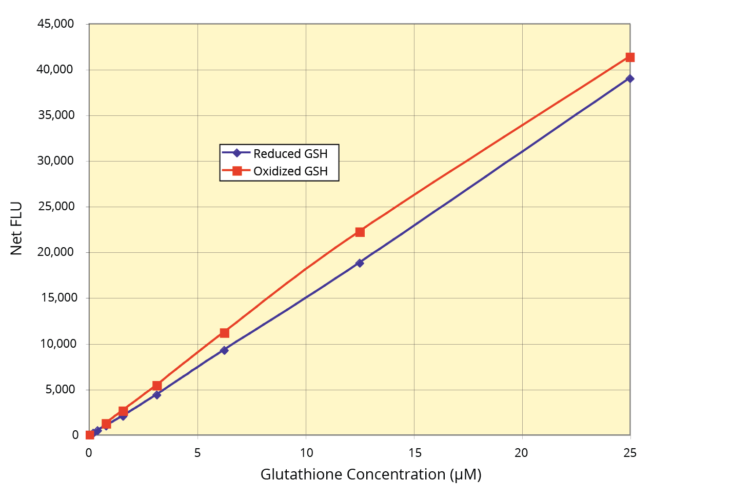
The DetectX® Glutathione (GSH) Fluorescent Detection Kit (384-Well Plate) provides a precise method for measuring GSH levels in various samples. These include whole blood, serum, plasma (EDTA and Heparin), erythrocytes, urine, cell lysates, and tissue. This kit features a short run time of only 15 or 30 minutes. Users should consult the detailed kit insert for full instructions. The kit includes a Glutathione standard for establishing an accurate standard curve.
Protocol Summary:
- Transfer standards or diluted samples into the black microtiter plate.
- Add ThioStar® Detection Reagent to each well, ensuring even mixing.
- (Optional) Incubate the mixture at room temperature for 15 minutes. Use a plate reader to measure the fluorescent signal to calculate the Free GSH concentration.
- Add Reaction Mixture to each well and incubate at room temperature for 15 minutes.
- After incubation, use a plate reader to measure the fluorescent signal. Calculate the Total GSH concentration using the standard curve.
Background:
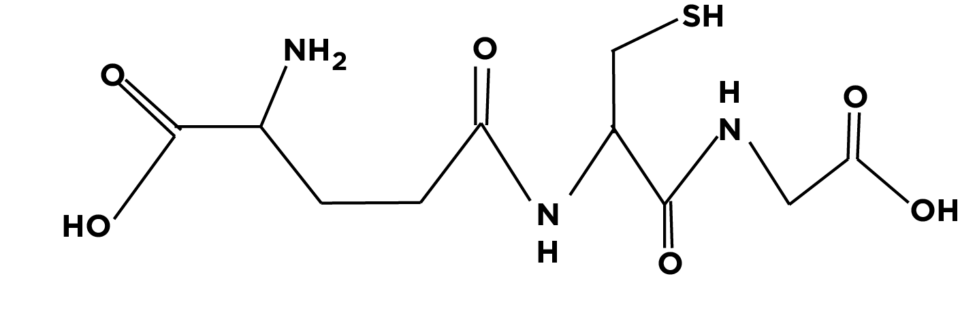
Glutathione (L-γ-glutamyl-L-cysteinyl glycine; GSH) is a critical non-protein thiol found in mammalian cells, with concentrations typically ranging from 0.5 to 10 mM. As a tripeptide, GSH is composed of glutamate, cysteine, and glycine. It plays a key role as an antioxidant, protecting cellular components from oxidative damage.
GSH undergoes enzymatic conversion to its oxidized form, glutathione disulfide (GSSG). In its role as an electron donor, GSH is instrumental in reducing disulfide bonds in cytoplasmic proteins to cysteines. The prevalence of its reduced form in cells is due to the activity of glutathione reductase, which is both constitutive and inducible in response to oxidative stress.
Researchers commonly use the ratio of reduced to oxidized Glutathione within cells as a metric for assessing cellular toxicity. This ratio reflects the redox state of the cell and is crucial in studies of cellular response to oxidative stress and toxicity. Therefore, accurate measurement of both reduced and oxidized forms of Glutathione is essential in research areas such as toxicology, pharmacology, and the study of various diseases where oxidative stress plays a role.
The DetectX® Glutathione (GSH) Fluorescent Detection Kit (384-Well Plate), with its efficient and sensitive detection system, is an important tool in these research fields, enabling precise measurement of GSH levels and contributing to our understanding of cellular health and disease mechanisms.