Allopregnanolone ELISA Kit
Personal Touch
Here to Help.
Ready to Ship
Most kits in stock.
Easy to Use
Simple protocols.
- Catalog Number K061-H
- Assay Type Competitive ELISA
- Sample Types Urine, Extracted Serum, Extracted Plasma (EDTA and Heparin), Dried Feces, Tissue Culture Media
- Sensitivity 129.7 pg/mL
- Species Allopregnanolone is identical across species
- Assay Duration 2.5 Hours or Overnight
- Samples/Plate 39 in Duplicate
- Readout Colorimetric, 450 nm
Assay Principle:
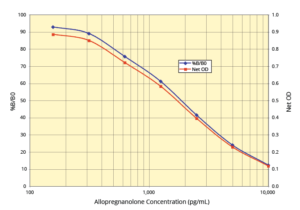
The DetectX® Allopregnanolone ELISA Kit quantitatively measures allopregnanolone in urine, extracted serum, extracted plasma (EDTA and Heparin), dried feces, and tissue culture media. This competitive ELISA has a run time of 2.5 hours. Please read the complete kit insert for more information before performing this assay. Use our provided allopregnanolone standard to generate a standard curve for the assay.
Protocol Summary:
- Add standards or diluted samples to the provided transparent microtiter plate coated with goat anti-mouse IgG antibody.
- Introduce the allopregnanolone peroxidase conjugate and allopregnanolone mouse monoclonal antibody to initiate the immunological reaction.
- Incubate the plate covered, shaking at room temperature for 2 hours. The reaction varies inversely with the allopregnanolone concentration in the sample.
- After incubation, remove excess conjugate and add the TMB substrate. The substrate reacts with the bound conjugate to produce a detectable signal.
- Measure the signal intensity at 450nm with a plate reader and calculate allopregnanolone concentration using the standard curve.
Background:
Allopregnanolone, also known as 3α-hydroxy-5α-pregnan-20-one, THP, or THPROG, is a significant neurosteroid in blood and brain tissues. It is a metabolite of progesterone and a powerful modulator of GABAA receptors, exhibiting notable anxiolytic and anticonvulsant properties. Allopregnanolone biosynthesis involves converting progesterone into 5α-dihydro progesterone by 5α-reductase type I, followed by conversion into allopregnanolone by 3α-hydroxysteroid oxidoreductase isoenzymes.
Allopregnanolone’s impact extends to neurological health, with links to conditions like anxiety and depression, particularly when its synthesis is inhibited by drugs such as finasteride and dutasteride. Its role in neurogenesis is also significant, with studies showing potential in reversing neuron deficits and cognitive impairments in Alzheimer’s disease models. This highlights allopregnanolone’s importance in both physiological and pathological contexts, particularly in neurology and mental health.
The DetectX® Allopregnanolone ELISA Kit is a tool for researchers in neurology and endocrinology, enabling the accurate measurement of allopregnanolone levels in various biological samples. This kit provides valuable insights into the neurosteroid’s role in various physiological processes and disease states.