Formaldehyde Fluorescent Detection Kit
- Catalog Number K001-F1
- Assay Type Detection Kit
- Sample Types Urine, Tissue Culture Media
- Sensitivity 0.715 μM
- Species Formaldehylde is identical across species
- Assay Duration 30 Minutes
- Samples/Plate 40 in Duplicate
- Readout Fluorescent, 510 nm emission / 450 nm excitation
Personal Touch
Here to Help.
Ready to Ship
Most kits in stock.
Easy to Use
Simple protocols.
Assay Principle:
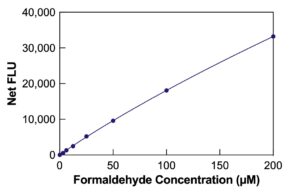
The Formaldehyde Fluorescent Detection Kit efficiently quantifies formaldehyde in urine, water, and tissue culture media with a quick 30-minute run time. For detailed instructions, refer to the complete kit insert. Generate a standard curve for the assay using the provided formaldehyde standard.
Protocol Summary:
- Add samples or standards to the black microtiter plate.
- Mix with DetectX® Formaldehyde Reagent, and incubate at 37°C for 30 minutes.
- Measure the fluorescent signal with a plate reader to determine formaldehyde concentration.
Background:
Formaldehyde, a colorless, flammable gas, is significant in the study of oxidative stress and metabolomics. As both a byproduct and a contributor to oxidative stress, it plays a dual role in biological systems. Oxidative stress, characterized by an imbalance between reactive oxygen species and antioxidants, leads to cellular damage. Formaldehyde, whether endogenously produced or environmentally introduced, can exacerbate this imbalance. Its detection is pivotal in researching its correlation with conditions like aging, neurodegeneration, and cardiovascular diseases.
Formaldehyde is also a key metabolite, with variations in its levels indicating shifts in metabolic pathways. It’s essential in studies focused on toxicology, pharmacology, and disease pathology, where understanding metabolome changes is crucial.
Environmental exposure to formaldehyde impacts human health by altering oxidative stress responses and metabolic pathways. Accurate measurement of its levels is vital for assessing its effects on human health, particularly in linking environmental factors to metabolic disorders.
The Formaldehyde Detection Kit is therefore crucial in environmental monitoring and biomedical research, helping to elucidate formaldehyde’s role in oxidative stress and metabolome variations.
Featured Publications:
1. McKenzie, R. M. E., et al. (2015). “Metabolome variations in the Porphyromonas gingivalis vimA mutant during hydrogen peroxide-induced oxidative stress.” Molecular Oral Microbiology 30(2): 111-127.
2. Neuss, S., et al. (2010). “Exposure of human nasal epithelial cells to formaldehyde does not lead to DNA damage in lymphocytes after co-cultivation.” Mutagenesis 25(4): 359-364.