Palladium API Screening Fluorescent Detection Kit
- Catalog Number K007-F1
- Assay Type Detection Kit
- Sample Types Active Pharmaceutical Ingredients
- Assay Duration 30 Minutes
- Samples/Plate 41 in Duplicate
- Readout Fluorescent, 520 nm Emission / 485 nm Excitation
Personal Touch
Here to Help.
Ready to Ship
Most kits in stock.
Easy to Use
Simple protocols.
Assay Principle:
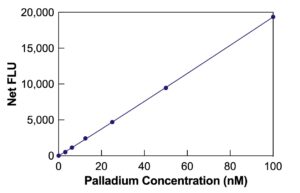
The DetectX® Palladium API Screening Fluorescent Detection Kit offers a reliable and efficient method for measuring Palladium levels in API scavenging steps. The kit provides a straightforward process with a run time of 30 minutes. Please read the kit insert for comprehensive instructions. A Palladium standard is included to create an accurate standard curve for the assay.
Protocol Summary:
- Add standards or diluted samples into the black microtiter plate.
- Introduce PdX™ Palladium Detection Reagent and Sodium Borohydride Reagent solution into each well, mixing completely.
- Incubate the mixture at room temperature for 30 minutes, enabling the fluorescent reaction between the PdX™ Reagent and reducing Palladium in the sample.
- After incubation, use a plate reader to detect and measure the fluorescent signal, and calculate Palladium concentration using the standard curve.
Background:
Palladium (Pd) compounds are extensively used in pharmaceutical processes, particularly for catalyzing carbon-carbon and carbon-heteroatom coupling reactions. These reactions are fundamental in creating complex molecules using various functional groups. However, a challenge arises as palladium may remain in the isolated active pharmaceutical ingredient (API) product, raising concerns about contamination.
Regulatory bodies like the FDA and EMEA have set stringent limits on the contamination of platinum group metals (including Palladium) in pharmaceutical products, typically capping it at less than 5 ppm. Traditional methods for evaluating Palladium in APIs involve costly equipment and require skilled personnel. These conventional techniques also face issues with probe cross-contamination and necessitate meticulous cleanup procedures to ensure accuracy.
The DetectX® Palladium API Screening Kit addresses these challenges by providing a more accessible and efficient method for Palladium quantification. This kit reduces the need for expensive instrumentation and extensive scientist training, simplifying the process of ensuring regulatory compliance in pharmaceutical manufacturing. Its sensitive detection system enables accurate Palladium measurement, crucial for maintaining the purity and quality of API products.