Can PGE2 Help Repair A Broken Spine?
A spinal cord injury (SCI) may occur due to a sudden blow to the spine causing vertebral fractures, compressions or dislocations. An SCI can also damage the spinal cord itself, which is a collective bundle of nervous tissues, causing muscle and body coordination issues and sometimes paralysis.
When the spinal cord is damaged, a strong inflammatory response is initiated by the central nervous system to stimulate the flow of blood and immune cells to the damaged area. Neuronal cells and white blood cells in injured nerves act together to release inflammatory molecules. This causes vasodilation and increased blood flow, thereby extending the inflammation and damage to the rest of the spinal cord. Some of these inflammatory molecules can recognize patterns and continue the inflammation and damage to neurons even after an injury. This leads to poor recovery and continued destruction of spinal nerve tissue. Some neuronal cells, such as astrocytes, secrete both inflammatory and anti-inflammatory markers to fine tune and prevent inflammation from spreading to other parts of the body. Recently, these anti-inflammatory markers have gained interest as possible SCI therapeutics, since they have the potential to reduce inflammation and prevent prolonged damage to spinal neurons.
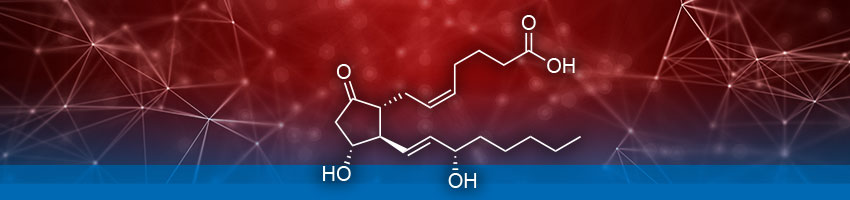
Previous studies have independently identified macrophage migration inhibitory factor (MIF) and Prostaglandin E2 (PGE2) as two molecules capable of fine-tuning inflammation during SCI. MIF is known to either activate or diffuse the inflammation based on environmental cues. PGE2 also exhibits both pro- and anti-inflammatory properties. In a 2018 paper, Zang et al., explores the association of MIF and PGE2 in astrocyte-mediated fine-tuning of cellular homeostasis during SCI with the aim of discovering a therapeutic intervention pathway.
In the study, experiments were done using SCI and control rat models. Astrocyte neuronal cells were isolated and analyzed using western blotting to qualitatively assess if MIF facilitates PGE2 synthesis. The Arbor Assays DetectX® PGE2 Enzyme Immunoassay (ELISA), K051-H1/H5, was used to quantify PGE2 level. Further western blotting, immunohistochemistry, qPCR, and ELISA experiments were performed to identify and quantify other proteins involved in cellular pathways regulating the production of PGE2 by MIF. Evaluation of neuronal cells treated with various inhibitors was also carried out to validate the predicted enzymatic pathway.
Western blotting, ELISA and qPCR experiments demonstrated that MIF stimulates PGE2 synthesis during SCI through the activation of the mitogen-activated protein kinases (MAPKs) pathway. Additional western blotting and inhibitory experiments showed that overproduction of PGE2 regulates pro-inflammatory markers by either downregulation or upregulation. For example, results demonstrated that PGE2 downregulated TNF-α and upregulated interleukins to maintain neuronal homeostasis.
To summarize, this study showed a novel mechanism of action of MIF and PGE2 in fine-tuning neuronal inflammation during SCI and unveiled biomarkers for managing SCI-related neuronal inflammation for therapeutic intervention.
