Protein Kinase A (PKA) Colorimetric Activity Kit
- Catalog Number K027-H1
- Assay Type Activity Assay
- Sample Types Cell Lysates, Tissue Extracts, Purified Systems
- Sensitivity 0.037 Units/mL
- Species Identical across all species
- Assay Duration 3 Hours
- Samples/Plate 42 in Duplicate
- Readout Colorimetric, 450 nm
Personal Touch
Here to Help.
Ready to Ship
Most kits in stock.
Easy to Use
Simple protocols.
Assay Principle:
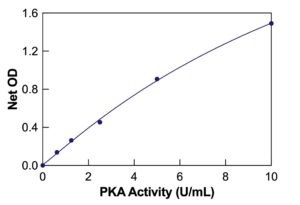
The DetectX® Protein Kinase A (PKA) Colorimetric Activity Kit provides an efficient and precise method for quantitatively measuring PKA activity in cell lysates, tissue extracts, and purified systems. This activity assay has a run time of 3 hours. Users should thoroughly read the complete kit insert for detailed instructions before performing the assay. The kit includes a PKA Standard to establish an accurate standard curve.
Protocol Summary:
- Add standards or diluted samples in kinase assay buffer to the provided transparent microtiter plate with bound PKA substrate.
- Introduce reconstituted ATP to each well, ensuring proper mixing of reagents.
- Incubate the sealed plate at 30°C with shaking for 90 minutes.
- Add Donkey anti-Rabbit IgG HRP Conjugate and Rabbit Phospho PKA Substrate Antibody to each well. Incubate at room temperature for 60 minutes.
- After incubation, wash away excess phospho-PKA-peroxidase conjugate. Then add the TMB substrate, which reacts with the bound conjugate to generate a detectable signal.
- Use a plate reader to measure the signal at 450nm and calculate PKA activity using the standard curve.
Background:
Protein Kinase A (PKA) is activated when cyclic AMP (cAMP) levels rise following stimulation of G Protein-coupled receptors and adenylyl cyclase. The expressed PKA holoenzyme comprises two catalytic (C) and two regulatory (R) subunits. In its inactive state, the pseudosubstrate sequences on the R subunits inhibit the activity of the C subunits, which are released as active monomers upon cAMP binding to the R subunits. PKA’s localization and interaction with A kinase-anchoring proteins (AKAPs) regulate the phosphorylation of specific substrates, including Bad (Ser155), CREB (Ser133), and GSK-3 (GSK-3α Ser21 and GSK-3β Ser9). PKA shares substrate specificity with Akt (PKB) and PKC.
PKA plays a vital role in various physiological processes, including cancer progression, vasodilation, and metabolic regulation. It is particularly significant in cardiac function and remodeling and shows substantial alterations in signaling in conditions like obesity. The DetectX® Protein Kinase A (PKA) Colorimetric Activity Kit, with its high sensitivity and specificity, is an important tool for researchers studying the diverse roles of PKA in health and disease.