Hydrogen Peroxide (H2O2) Fluorescent Detection Kit
- Catalog Number K034-F1
- Assay Type Detection Kit
- Sample Types Urine, Buffer, Tissue Culture Media
- Sensitivity 0.038 µM
- Species All Species
- Assay Duration 15 Minutes
- Samples/Plate 40 in duplicate
- Readout Fluorescent, 590 nm Emission / 520 nm Excitation
Personal Touch
Here to Help.
Ready to Ship
Most kits in stock.
Easy to Use
Simple protocols.
Assay Principle:
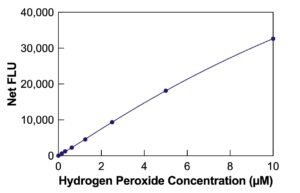
The DetectX® Hydrogen Peroxide (H2O2) Fluorescent Detection Kit quantitatively measures H2O2 levels in urine, buffer, and tissue culture media. With a run time of only 15 minutes, this kit provides accurate and efficient analysis. Read the complete kit insert thoroughly before starting the assay. The kit includes a Hydrogen Peroxide standard for establishing an accurate standard curve.
Protocol Summary:
- Introduce standards or diluted samples into the provided black microtiter plate.
- Add Fluorescent Detection Reagent and Horseradish Peroxidase Concentrate to each well, ensuring proper mixing.
- Incubate the mixture at room temperature for 15 minutes. The fluorescent reaction occurs between the detection reagents and H2O2 in the sample.
- After incubation, use a plate reader to detect the generated fluorescent signal at 590nm. Calculate H2O2 concentration using the intensity and the standard curve.
Background:
Hydrogen peroxide (H2O2) is a significant reactive oxygen species in biological systems. It is produced from the incomplete reduction of O2 during respiration. This process generates superoxide anions (O2 -·), and subsequently H2O2 through dismutation. Low levels of O2 -· and H2O2 are created in response to various extracellular stimuli, including cytokines, peptide growth factors, GPCR agonists, and shear stress.
The Fenton reaction, described in 1894, involves the oxidation of substances by Fe2+ and H2O2. Exogenous or intracellularly produced H2O2 in response to receptor stimulation can impact various proteins, including kinases, phosphatases, transcription factors, and ion channels. H2O2, along with O2, may also participate in generating reactive species like singlet oxygen and peroxynitrite, especially in reactions involving iron, contributing significantly to H2O2 toxicity under certain conditions.
Measuring hydrogen peroxide (H2O2) in urine, buffer, and tissue culture media is essential for evaluating oxidative stress and its impact on systemic metabolism, biochemical processes, and cellular responses. H2O2 quantitation is particularly important in studies focused on aging, cancer, and neurodegeneration. The DetectX® Hydrogen Peroxide (H2O2) Fluorescent Detection Kit is a valuable tool for researchers studying oxidative stress, cellular signaling mechanisms, and the effects of reactive oxygen species in various biological contexts.