Confusing P450 Data? Simply Measure Formaldehyde
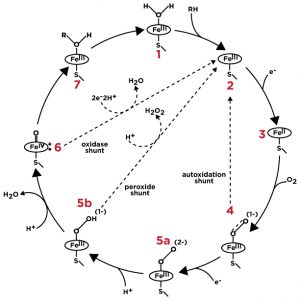
The cytochrome P450 (CYP) superfamily of heme-containing enzymes displays great diversity with regard to substrate specificity and catalytic activity. P450s use a plethora of both exogenous and endogenous compounds as substrates in enzymatic reactions, often functioning as the terminal oxidase enzymes in electron transfer reactions (see figure below). Catalysis by eukaryotic P450 enzymes typically involves a multistep reaction cycle that includes two steps in which electron transfer is accomplished from a redox partner. In humans P450 enzymes are primarily membrane associated proteins found in the endoplasmic reticulum and mitochondria in most tissues of the body. They play important roles in hormone synthesis and breakdown, cholesterol synthesis, and vitamin D metabolism as well as the metabolism of potentially toxic compounds such as drugs and bilirubin. Analysis of P450 enzymes can play a crucial role in the development of new drug entities as drug-drug interactions commonly arise from the inhibition of cytochrome P450 activities.
P450 Catalytic Cycle
Many drugs increase or decrease the activity of various CYP isozymes either by inducing the biosynthesis of an isozyme (enzyme induction) or by directly inhibiting the activity of the CYP (enzyme inhibition). These effects can be a major source of adverse drug interactions, since changes in CYP enzyme activity may affect the metabolism and clearance of various drugs. For example, if one drug inhibits the CYP-mediated metabolism of a second drug, the second drug may accumulate within the body to toxic levels. Conversely, if the first drug increases the CYP-mediated metabolism of the second drug it may clear too quickly and therapeutic levels may not be maintained between doses. Such drug interactions may necessitate dosage adjustments or modifying treatment protocols to choose drugs that do not interact with the CYP system. These drug interactions are especially important to take into account when using drugs of vital importance to the patient, drugs with critical side-effects, and drugs with small therapeutic windows. However, any drug may be subject to an altered plasma concentration due to altered drug metabolism.
Our P450 Assay
Similar to the effect of drugs on P450 systems, the addition of a reporter compound may inhibit or induce P450 enzyme activity. Our P450 Demethylating Fluorescent Activity (K011-F1) kit measures the amount of formaldehyde produced by the action of P450 on its substrate rather than using a reporter substrate. This unique design means that no additions are made to the P450-substrate reaction. Carry out your P450-substrate reaction in the plates supplied with our kit and allow incubation under your normal reaction conditions. At the end of your reaction, add the Formaldehyde Detection Reagent to each well, incubate for 30 minutes and measure the formaldehyde produced by your P450 system by fluorescence (Ex. 450 nm / Em. 510nm). We supply a Formaldehyde standard to allow cross lab quantitation.
Advantages:
- Measure P450 activity without introducing a reporter compound or other additions to the native reaction
- Works with any demethylating P450 system
- Calibrated to formaldehyde standard
- Simple fluorescent results without the need for radioactivity or LC-MS
- Superior 4°C liquid stability
Related product
Formaldehyde Fluorescent Detection Kit
Publication
| Jan 01 2013 | Development, characterization, and use of a porcine epiblast-derived liver stem cell line: ARS-PICM-19 |