Simplify your Glutathione Measurements
Glutathione (GSH) is a tripeptide (gamma-L-glutamyl-L-cysteinylglycine) that is found in all mammalian tissues and is especially concentrated in the liver (1-10 mM). Along with its known function combating oxidative stress, GSH is also important for the detoxification of xenobiotics and metabolites, the regulation of cell growth and cell death, the regulation of the nitric oxide cycle, the storage of cysteine, the maintenance of redox potential and the modulation of immune function.
GSH is synthesized in the cytosol in two ATP-dependent steps. The first step is the unusual coupling of the gamma-carboxylic acid of glutamic acid to cysteine by the enzyme gamma-glutamylcysteine synthase to form gamma-glutamylcysteine. This is followed by the addition of glycine to the C-terminus of gamma-glutamylcysteine via the enzyme GSH synthetase to form GSH. The formation of gamma-glutamylcysteine is the rate-limiting reaction in GSH synthesis and is feedback inhibited by GSH itself, a mechanism responsible for the regulation of cellular GSH concentration. Because the unusual bond formed through the gamma-carboxyl group of glutamate can only be hydrolyzed by one enzyme, gamma-glutamyltranspeptidase, which is present only on the external surfaces of certain cells; GSH is highly resistant to intracellular degradation.
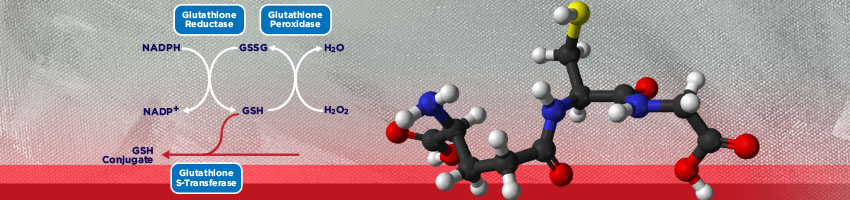
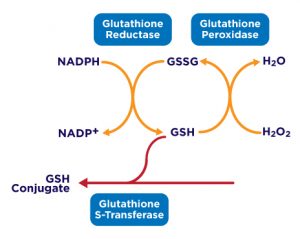
Glutathione exists in both the thiol-reduced (GSH) form and the disulfide-oxidized (GSSG) form. In the reduced state, the thiol group of cysteine is able to donate an electron to other molecules. The electron donation can affect the receiving molecule in varying ways including neutralizing reactive oxygen species, or maintaining cysteine in proteins in their reduced forms. When donating an electron, glutathione itself becomes reactive and readily reacts with another reactive glutathione to form GSSG. GSH can be regenerated from GSSG by the enzyme glutathione reductase. Although over 90% of glutathione exists in the GSH form, a true assessment of total glutathione concentration requires the measurement of both GSH and GSSG. The ratio of GSH to GSSG can be changed by a number of factors including disease state or oxidative stressors.
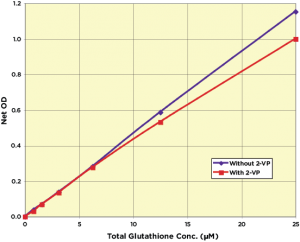
The Arbor Assays DetectX Glutathione Colorimetric Detection Kits (K006-H1) offers a quantitative method capable of measuring GSH in a variety of samples using a colorimetric substrate that yields a highly colored product. Free GSH concentration in the sample is calculated from the difference between two separate measurements – the total GSH measurement and a measurement of the GSH generated from GSSG. The kit reagents are stable at 4°C and sufficient for one 96-well plate. Sensitivity is less than 65 nM of GSH.
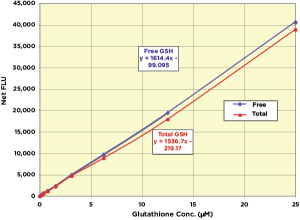
The Arbor Assays DetectX Glutathione Fluorescent Detection Kits (K006-F) offer a supersensitive method for measuring GSH in a variety of samples using ThioStar®. In this method Free and Total GSH are measured in the same well, with only one sample required for both results. Kit reagents are stable at 4°C and available as either one 96-well plate, five 96-well plates, or two 384 well plates. Sensitivity is less then 50 nM for both Free and Total GSH.
See More Oxidative Stress Products