mTOR Signaling and Podocyte Injury: 14 Years of Insight Into Diabetic Nephropathy Research
Diabetic nephropathy remains one of the most devastating long-term complications of diabetes, driving progressive kidney failure despite advances in glycemic control. While early research focused mainly on systemic metabolic stress, a pivotal 2011 study fundamentally reshaped the field by identifying podocyte-specific signaling events as a primary driver of disease progression.
In the highly cited Journal of Clinical Investigation paper, Inoki et al. demonstrated that aberrant activation of mTOR complex 1 (mTORC1) in podocytes is not merely associated with diabetic nephropathy; it is sufficient to cause it. More than a decade later, this work continues to anchor mechanistic and translational research in diabetes-related kidney disease.
Linking Molecular Signaling to Functional Kidney Damage
Using genetically engineered mouse models, the researchers selectively activated mTORC1 signaling in podocytes by deleting Tsc1, a key upstream negative regulator. This targeted approach allowed them to isolate the effects of podocyte-specific mTORC1 activation from systemic metabolic changes.
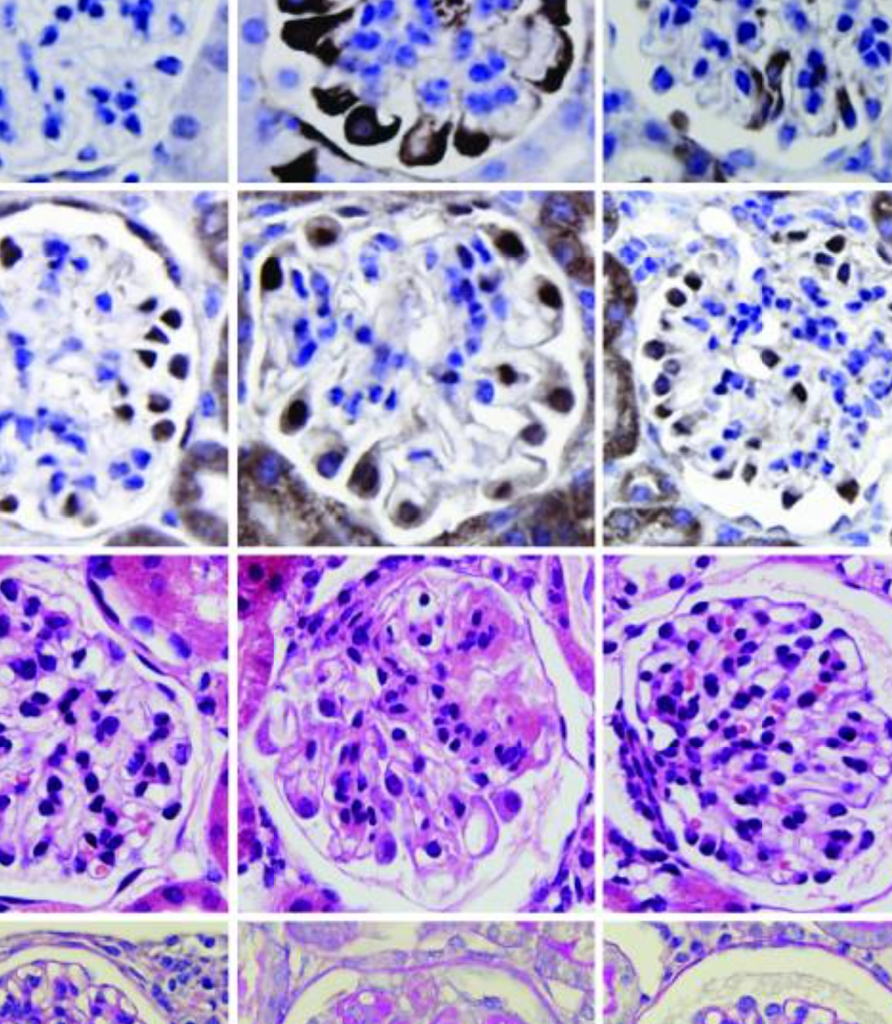
Across multiple models and time points, the study showed that sustained mTORC1 activation led to hallmark features of diabetic nephropathy, including:
- Podocyte foot process effacement
- Mesangial expansion and glomerular basement membrane thickening
- Progressive proteinuria and declining renal function
Importantly, partial inhibition of mTORC1 activity (either genetically or pharmacologically with rapamycin) significantly mitigated these effects, reinforcing the idea that precise regulation, rather than complete pathway suppression, is critical for kidney health.
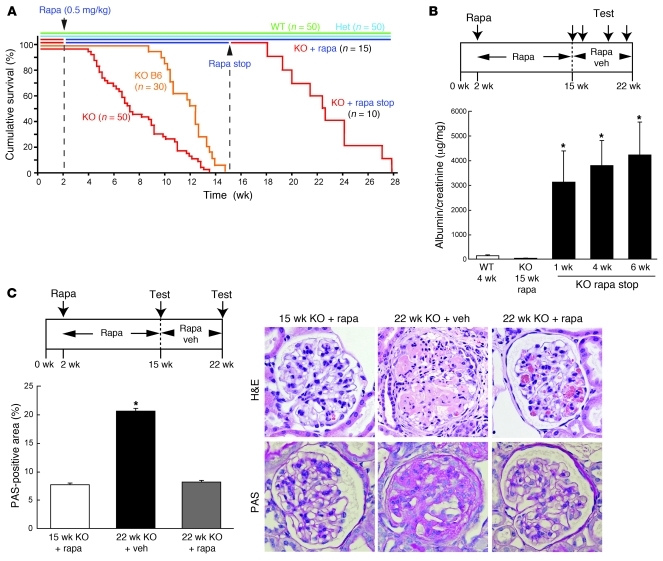
Figure. Albumin/creatinine reveals progressive renal failure driven by podocyte-specific mTORC1 activation. PcKOTsc1 mice exhibit significantly elevated biomarker concentrations, indicating impaired kidney function associated with sustained mTORC1 signaling. Quantitative ELISA-based measurement of urine and serum provided a functional assessment of disease severity in this diabetic nephropathy mouse model. Figure from Inoki K. et al., Journal of Clinical Investigation, 2011.
Quantitative Biomarkers in Mouse Models of Diabetes
A defining strength of this study is its integration of molecular mechanisms with functional disease endpoints. Rather than relying on histology alone, the authors used quantitative biomarkers to track kidney injury and recovery over time.
Key measurements included urinary albumin and creatinine to assess proteinuria; serum creatinine, measured using the DetectX® Serum Creatinine Kit (KB02-H), to evaluate renal function; and longitudinal comparisons across genetic backgrounds and treatment conditions.
These measurements depend on highly reproducible assay performance, especially in chronic disease models where subtle changes accumulate over weeks or months. In this study, ELISA-based quantification enabled clear differentiation between disease progression, therapeutic response, and genetic rescue.
Reliable Measurements Stand the Test of Time
Fourteen years on, this study remains influential because its conclusions were built on dependable measurements. By pairing precise mouse genetics with quantitative biomarkers such as serum creatinine, the authors directly linked molecular signaling to functional renal decline.
In chronic disease models such as diabetic nephropathy, where changes accumulate gradually, consistent assay performance is essential. Reliable biomarker quantification enables comparison of results across time, models, and laboratories.
Explore Arbor Assays’ publication database to see how researchers continue to rely on validated assays to support reproducible preclinical and translational research.
Featured Products
-
In Stock
Serum Creatinine Detection Kit
Price range: $321.00 through $514.00The DetectX® Serum Creatinine Kits measure creatinine in serum and plasma samples.