Serum vs Plasma: Does Your Sample Choice Matter?
A review of the literature quickly reveals that most biomarker studies rely on serum or plasma samples. In many cases, that choice appears so routine it’s rarely discussed. As one researcher jokingly put it, sometimes the deciding factor is simply whether EDTA tubes were already stocked in the lab.
While existing workflows and available supplies do influence experimental design, the choice between serum and plasma is rarely arbitrary. Each matrix reflects a slightly different biological state and introduces distinct considerations for sample handling, analyte stability, and data interpretation. Understanding these differences helps researchers select a sample type that aligns with their hypothesis.
Serum and Plasma Dominate Biomarker Research for Good Reason
Serum and plasma are the most commonly used sample types in biomarker research because they provide a direct, system-level snapshot of physiology. Circulating blood integrates signals from multiple organs and tissues, making it an efficient matrix for measuring hormones, cytokines, metabolites, and other soluble analytes linked to disease, stress, and homeostasis.
From a practical standpoint, serum and plasma are also well suited to experimental workflows:
- Blood collection is relatively standardized across species and study types
- Sample volumes are typically sufficient to support multiplexed or longitudinal analyses
- Extensive historical data enables cross-study comparison and replication
For these reasons, many publications that use Arbor Assays Kits measure analytes in serum or plasma, particularly in translational and pre-clinical models where reproducibility and comparability are essential.
What’s the Difference Between Serum and Plasma?
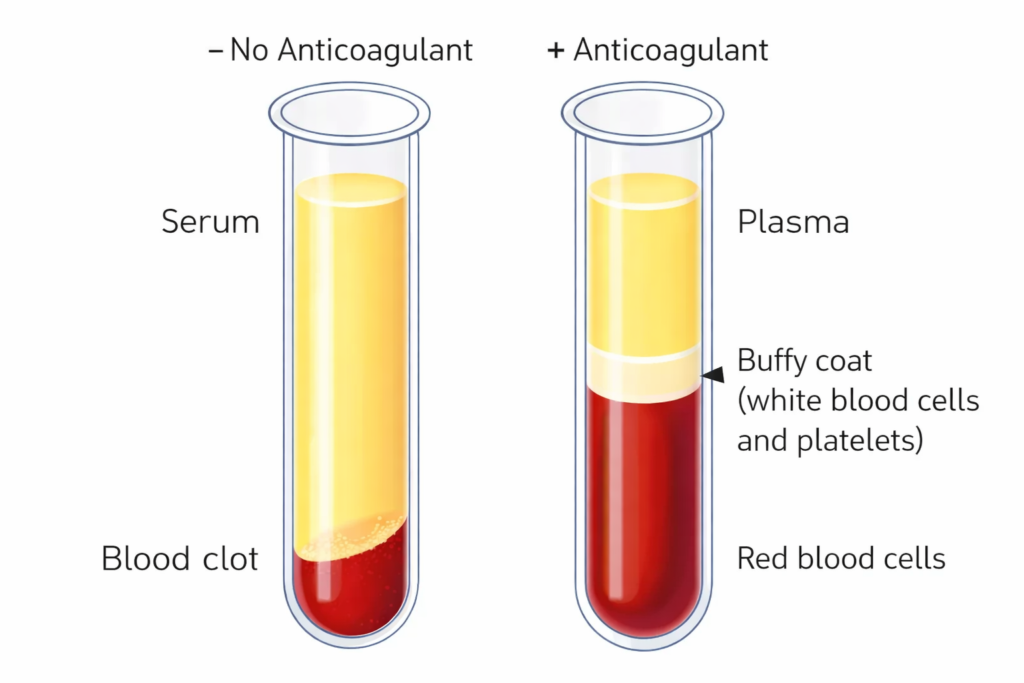
Although serum and plasma are both derived from whole blood, they represent distinct biological states shaped by the manner in which the sample is collected and processed.
Serum
Serum is collected after blood is allowed to clot in the absence of anticoagulants. During coagulation, clotting factors are consumed, and platelets become activated, thereby releasing or redistributing certain biomolecules. While this process has little effect on many stable hormones and metabolites, it can influence concentrations of certain cytokines involved in inflammation, immunity, or stress response.
Serum is therefore commonly used for analytes that are clinically standardized or relatively stable through the clotting process, including:
- Steroid hormones (e.g., cortisol, testosterone, estradiol)
- Thyroid hormones (T3, T4, TSH)
- Metabolic markers (e.g., insulin, leptin)
- Lipids and lipid-associated biomarkers
- Growth factors (e.g. PDGF released during clotting process)
Because most clinical reference intervals are defined in serum, this matrix is often preferred when studies aim to align with clinical diagnostics, compare with historical datasets, or measure analytes that remain stable during clot formation.
Plasma
Plasma is obtained by collecting blood into tubes containing an anticoagulant (e.g., EDTA, heparin, or citrate) and then centrifuging the sample to remove cells. Because clotting is prevented, plasma retains fibrinogen and other clotting factors, thereby minimizing ex vivo activation. As a result, plasma often reflects the circulating biochemical state at the moment of collection.
Plasma is frequently preferred for analytes that are sensitive to clotting, platelet activation, or rapid post-collection changes, including:
- Cytokines and chemokines (e.g., IL-6, TNF-α, MCP-1)
- Complement proteins and coagulation-related factors
- Certain peptide hormones with short half-lives
- Acute inflammatory and immune signaling markers
- Vascular and endothelial biomarkers affected by platelet activation
For studies focused on acute physiology, immune activation, or rapid signaling dynamics, plasma can reduce artifactual changes introduced during clot formation and provide a more accurate snapshot of circulating biology.
Importantly, many analytes can be measured in either serum or plasma when assays are properly validated. However, matrix choice can influence baseline values, variability, and interpretability. Neither serum nor plasma is inherently superior; rather, each provides a unique biological snapshot. The appropriate choice depends on the physiological question being asked rather than convention alone.
Practical and Experimental Design Considerations
In real-world research settings, biological ideals must coexist with experimental logistics. Study design, available materials, and downstream applications often shape sample selection. Researchers must weigh several factors:
- Existing collection workflows: Many labs standardize blood draws across multiple assays or collaborators; therefore, sample type must stay consistent.
- Sample availability: Historical collection protocols often dictate the sample type for longitudinal studies. When biobanked samples are involved, researchers cannot change protocols partway through the study.
- Multiplexing needs: When multiple assays are run from a single sample, researchers may choose the matrix that best accommodates all planned measurements.
- Anticoagulant compatibility: EDTA, heparin, and citrate can interact differently with specific assays, influencing matrix selection.
The reality of choosing serum or plasma based on what is already in the freezer is common and often appropriate, provided the assay has been validated for that matrix and sample handling is consistent across experimental groups.
Choosing Intentionally and Measuring with Confidence
Selecting serum or plasma is less about finding a “better” matrix and more about aligning the choice of matrix with biological intent and study design. What matters most is making that decision deliberately and maintaining consistency across experimental groups.
Using assays validated for the selected matrix is equally critical. Matrix-specific performance can influence sensitivity, variability, and data interpretation, particularly for low-abundance or signaling-sensitive biomarkers.
Arbor Assays’ immunoassays are validated across serum, plasma, and a wide range of additional sample types, giving researchers the flexibility to choose the matrix that best fits their experiment while maintaining confidence in data quality.
Ready to get measuring? Explore our lineup of highly published assay kits.
Featured Products
-
In Stock
Cortisol ELISA Kit
Price range: $346.00 through $1,388.00The DetectX® Cortisol ELISA Kits quantitatively measure cortisol present in a variety of samples.
-
In Stock
Triiodothyronine (T3) ELISA Kit
Price range: $505.00 through $2,017.00Quantitatively measures T3 present in urine, extracted serum, plasma, fecal extracts, and tissue culture media samples. It measures total T3 in extracted serum, plasma and fecal extracts.
-
In Stock
Serum 17β-Estradiol ELISA Kit
Price range: $410.00 through $1,638.00The DetectX® Serum 17β-Estradiol ELISA Kits quantitatively measure free 17β-Estradiol in serum and plasma.