Organic Cation Transporter-Mediated Accumulation of Quinolinium Salts in the LV Myocardium of Rodents
Nuclear medicine traditionally uses single-photon emission computed tomography to perform myocardial perfusion imaging (MPI). Earlier studies have looked at C-11 labeled quaternary ammonium cations as potential MPI agents. In an April 2022 Molecular Imaging and Biology publication, Grievink H. et al. investigated if interactions of a newly designed radiopharmaceutical quinolinium salt-based positron emission tomography (PET) probe with organic cation transporters (OCTs) accounted for previously observed accumulation of quaternary ammonium cations in the left ventricle of rodents and swine. The hope is that if the accumulation is explained, it can be used to increase the clinical applicability of these new probes for PET-MPI.
Organic cation transporters (OCTs) are a class of plasma membrane transporters in the solute carrier 22 family, differentially expressed in tumors and cancer cell lines. In normal tissues, OCT1 is predominantly found in the liver tissue of humans and the liver and kidney tissues of rodents.
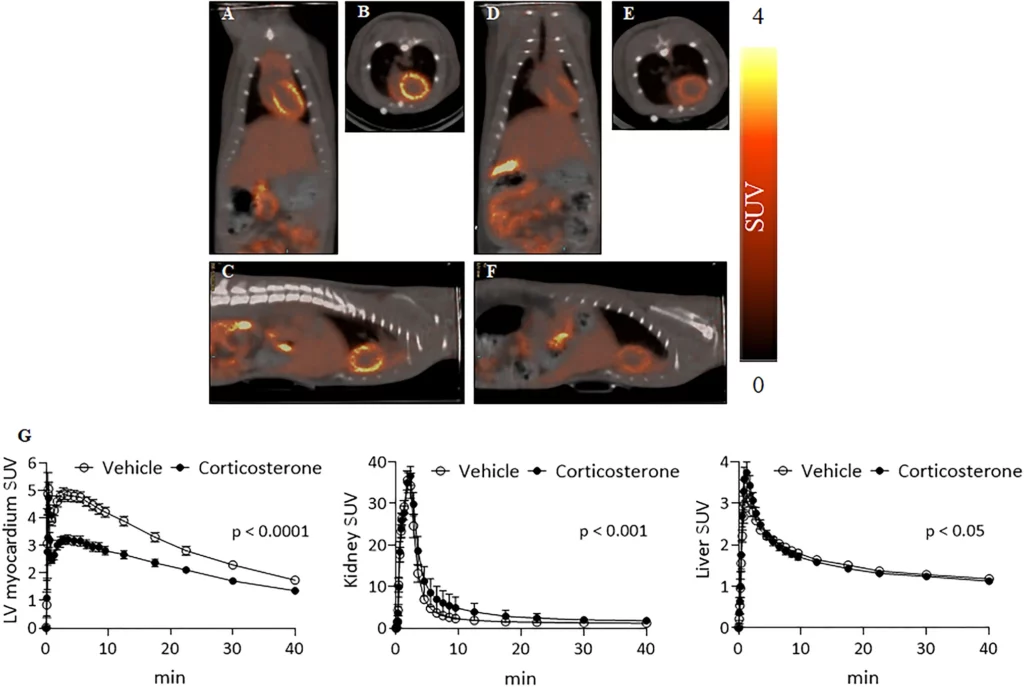
OCT2 is located in kidney tissue, while OCT3 has broader tissue distribution, found in the heart, skeletal muscle, liver, brain, and placenta tissues. OCTs mediate the absorption and secretion of organic cationic substances – endogenous compounds, various drugs, and xenobiotics. Inhibition can be achieved by non-transported compounds such as cations (decynium, d22), neural compounds (corticosterone), and anions (probenecid). The researchers studied the uptake and binding of the radiopharmaceutical in cancer cells transfected with human OCTs and evaluated PET imaging in tumor-inoculated rodents with and without pretreatment using d22 or corticosterone to inhibit the OCTs. Serum levels of corticosterone before radiopharmaceutical injection were measured using Arbor Assays DetectX® Corticosterone Multi-Format ELISA Kit, K014-H. As a final measure to test the findings from rodent to human, PET imaging was evaluated on a non-human primate to see if the accumulation results were the same as observed in the rodent model.
The cellular uptake studies using HEK293 cells highlighted the interaction of the radiopharmaceutical probe with hOCT1-3, with an increase in accumulation in the transfected cells compared to the control HEK293 cells. Additionally, the uptake was inhibited by corticosterone in a dose-dependent manner. Similar accumulation and inhibition were observed in the rodent model left ventricular myocardium. A single study in a monkey showed no apparent uptake of the radiopharmaceutical probe in the heart. In conclusion, the radiopharmaceutical probe was found to interact with OCTs, contributing at least in part to the accumulation in the left ventricular myocardium of rodents. The lack of accumulation seen in a non-human primate, which is most likely a low affinity, will limit the clinical potential as a PET pharmaceutical for human cardiac and/or OCT tumor screening.
Featured Products
-
In Stock
Corticosterone Multi-Format ELISA Kit
$366.00 – $1,462.00The DetectX® Corticosterone Multi-Format ELISA Kits quantitatively measures corticosterone present in a variety of samples.