NEW Levonorgestrel (LNG) Kit
Levonorgestrel (LNG) is a synthetic steroid analogue used in a number of birth control methods. Levonorgestrel was first made in the 1960s and has been used as a method of birth control since the 1980s. Today it is on the WHO List of Essential Medicines, and is available as a generic medication. LNG sold under the name “Plan B” can be used as an emergency birth control when taken within 120 hours. LNG may also be combined with an estrogen to make an oral contraceptive, or used alone as the mini-pill. As part of an LNG-releasing IUD, sold as Mirena among others, it is effective for long-term prevention of pregnancy. LNG works as a contraceptive by decreasing ovulation, changing cervix mucus, and altering the uterine lining.
Beyond contraception, levonorgestrel uses include treatment of heavy menstrual bleeding and dysmenorrhea. It can also be used as endometrial protection during estrogen replacement therapy in postmenopausal women. LNG has been shown to be an effective treatment modality for a variety of gynecologic conditions including idiopathic, myoma- or adenomyosis-related heavy menstrual bleeding, endometriosis- or adenomyosis-related pelvic pain, endometrial hyperplasia, and early-stage endometrial cancer. LNG binds to the progesterone receptor in the nucleus of target cells, resulting in a suppression of luteinizing hormone (LH) activity, an inhibition of ovulation, and alteration in the cervical mucus and endometrium.
Introducing our new Levonorgestrel (LNG) ELISA Kit
The DetectX Levonorgestrel (LNG) Immunoassay kit is designed to quantitatively measure levonorgestrel present in serum, plasma, saliva, urine, dried fecal samples, water, and tissue culture media samples. In extracted serum, plasma and fecal samples, the kit measures total LNG (free and bound).
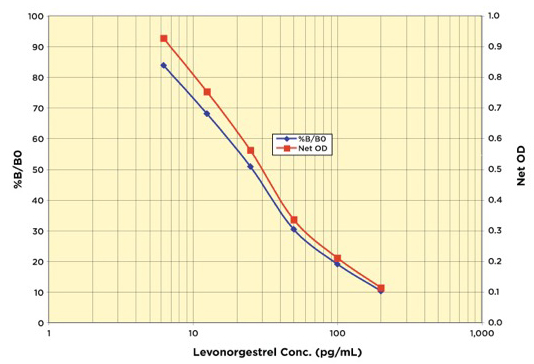
Featured Products
-
In Stock
Levonorgestrel (LNG) ELISA Kit
Price range: $696.00 through $2,781.00The DetectX® Levonorgestrel (LNG) ELISA Kits are designed to quantitatively measure Levonorgestrel present in serum, plasma, saliva, urine, dried fecal samples, milk, and tissue culture media samples.